
#Iso 14971 iso#
This class has been updated for ISO 14971:2019. Need to get smart about risk management in a hurry? Check out our intensive 3-day risk management training course. Identify hazards and estimate the probability that harm might occur.Įstimate the severity of each hazard and evaluate the associated risks.Ĭontrol the risks and monitor the effectiveness of the controls put in place. Review the intended use of your medical devices. The goal is to establish, document, and maintain a risk management process to: Oriel STAT A MATRIX can help you develop and implement the needed processes efficiently and correctly while providing a valuable “outsider perspective” on your medical device risk management practices. This level of risk assessment calls for careful planning and appropriate staffing to support the necessary activities. The standard requires you to review the risks posed by your devices over time and monitor any changes (e.g., design, customer input/feedback, and postmarket surveillance). Learn more about the updated ISO 14971.Ĭomplying with ISO 14971 can be challenging. A solid risk assessment program helps you identify design issues before distribution, eliminating dangerous problems and the costs associated with recalls.Īssessing your current compliance with ISO 14971:2019 ISO 14971 is an integral part of an effective quality management system and should be embedded into your medical device manufacturing life-cycle process.
#Iso 14971 software#
Interaction of software with its environment shall also be analyzed to ensure a complete risk assessment.ISO 14971 an international standard for risk management related to the manufacturing of medical devices.It is recognized by most regulatory authorities as the “de facto” standard for risk management. More, there are additional activities described in IEC 62304 that are required for software risk management. There is no bypass of any step described in the standard. To sum-up, the risk management process of ISO 14971 shall be applied as is to software. One symptom of a risk analysis that may not be accurate enough about interactions between hardware and software is the production of two separate risk assessment reports by the manufacturer, one for hardware and one for software. The main risk management process shall also contain risk management activities about interaction of software with hardware and its environment. On top of these two parts, risk analysis of software shall be realized with consideration to its environment. That software-related part should contain activities required by the IEC 62304 standard, like determining the software class (class A, b or C) and production of records, like software requirements mitigating risks and traceability matrixes. The part about software could be a work instruction that adds specific tasks to the main risk management procedure. One possible solution for manufacturers that design both hardware and software is to separate the risk management process in two parts, one for hardware and one for software. The risk assessment report is continuously updated during the software development process. The graph below shows that the risk management process during design has to be realized in parallel with the software development process.
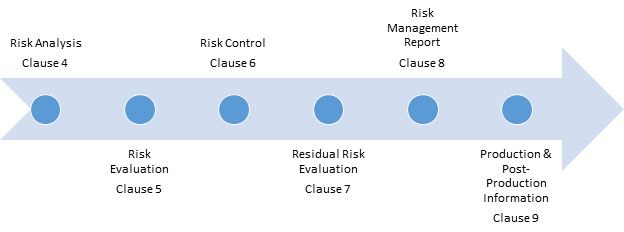
So design is the only way to mitigate risks (except some maintenance actions like archiving data or warnings in instructions for use, known to be of lesser efficiency). Mitigations actions for hardware may be replacing aging parts. But hazardous situations also happen because hardware wears, ages and breaks. One could argue this is also the case for hardware. The second main reason is the fact that mitigation actions are made by design. See for example my previous articles about risks, defects and software failures. The main reason of this situation is the difference in the causes and consequences of hazardous situations, and in the way they are detected, when software failure occurs. Activities of software (and other technical fields) can be seen as addendum to a main generic risk management process. The schema below represents the ISO 14971 risk management process as an area. That doesn’t mean that the ISO 14071 process has to be tweaked but that additional concepts and activities, mainly described in IEC 62304 standard, have to be implemented. When manufacturers design devices that embed software or are standalone software, a few peculiarities of software have to be integrated in the risk management process. The ISO 14971 standard describes a risk management process that medical devices manufacturers have to apply. Unlike ISO 13485 standard, there is no possible exclusion. The process described in ISO 14971 shall not work in fail-soft mode.
